Qlucore Insights Tests (RUO)
We work with scientific experts to develop disease specific tests for subtype determination. Machine learning (AI) based classifiers are combined with gene fusion detection and user-friendly visualizations. The tests also support different metrics for quality control.
- Acute lymphoblastic leukemia (BCP-ALL)
- Lung cancer with identification of metastatic origin
- Acute myeloid leukemia (AML)
- Urothelial carcinoma - bladder cancer
Read more about Qlucore as part of the EIC Accelerator precision oncology portfolio in Nature Biotechnology.
The Qlucore Insights Tests are for research use only (RUO). Development of tests for Qlucore Diagnostics (CE-mark) is ongoing and the test for BCP-ALL is available and certified under IVDR.
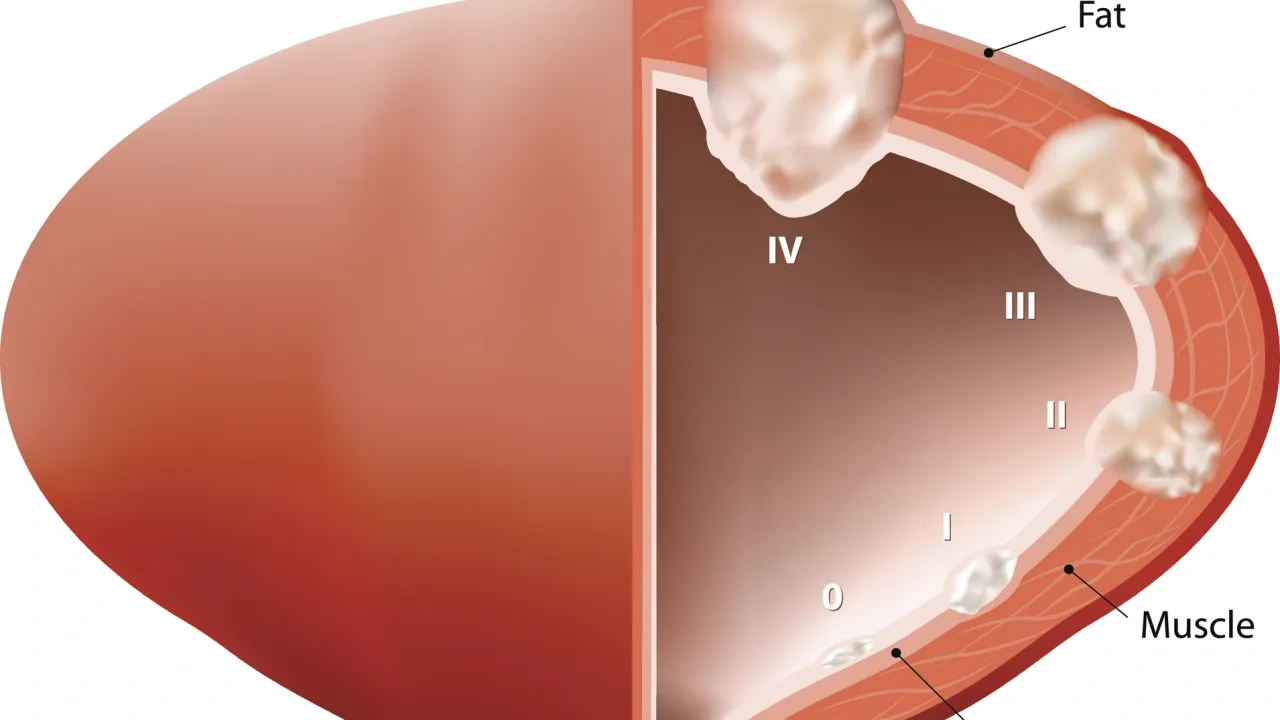
BCP-ALL (RUO)
Up to 7% more samples are covered compared to standard methods. The test is based on gene expression-based subtype classification in combination with comprehensive gene fusion analysis. With the inclusion of the DUX4-rearranged and ETV6::RUNX1-like subtypes, up to 7% more children are covered compared to standard methods. Supported subtypes:
1. High hyperdiploidy
2. ETV6::RUNX1 + ETV6::RUNX1-like
3. KMT2A(MLL)-rearranged (MLL)
4. TCF3::PBX1
5. BCR::ABL1 + BCR::ABL1-like
6. DUX4-rearranged
More information: BCP-ALL
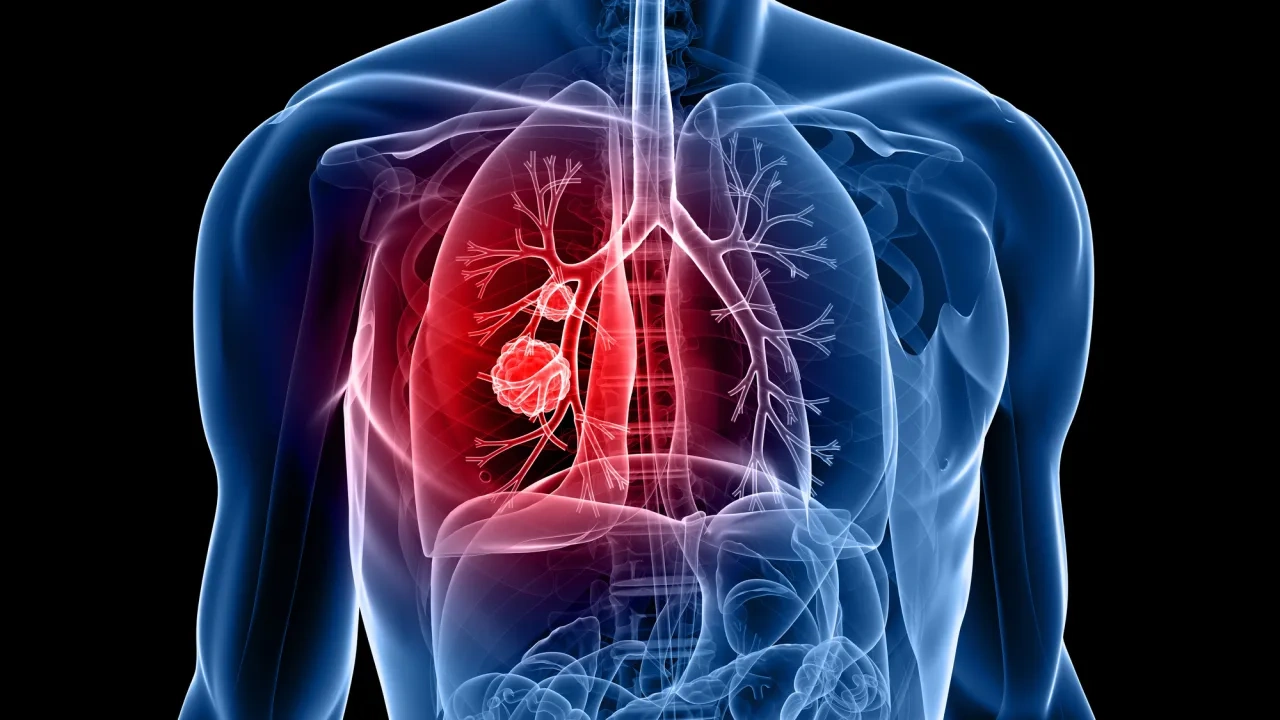
Lung cancer with detection of metastasis origin (RUO)
A cost efficient and complete solution for classification of lung lesions including detection of metastasis. The identification of the origin of metastasis early in the workup can be a significant cost saving. The lung cancer test provides a comprehensive overview of the lesion that is being analyzed. The sample tested is assigned to one of 19 subgroups based on the machine learning classifier. The subgroups include the major primary lung cancer subtypes, twelve subgroups for metastatic cancer such as breast, colorectal, or kidney cell cancer and two subtypes for infections/inflammatory conditions, i.e. sarcoidosis and TBC.
Gene fusion detection and analysis using up to three different gene fusion callers.
Sample: FFPE. From resection or biopsy.
More information regarding Lung Cancer.
AML (RUO)
Molecular subtype identification combined with cost efficient gene fusion detection for informed decision making. In addition, the overexpression of the MECOM gene is indicated as this enables a more comprehensive detection of samples that potentially should be classified as MECOM-r. The test lists all 30+ guideline mandated gene fusions in the report as well as novel gene fusions that match specified quality measurements.
The test detects the following subtypes:
- AML-MR
- CBFB::MYH11
- CEBPA bZIP
- MECOM-r
- KMT2A-r
- NPM1
- PML::RARA
- RUNX1::RUNX1T1
- TP53
Sample type: Fresh frozen
More information regarding AML.

Urothelial carcinoma - bladder cancer (RUO)
Avoid human uncertainties and improve risk scoring. The test is based on the molecular defined subtypes from the Lund Taxonomy and also include detection of the FGFR3(1&2) gene fusions as well as NTRK and RET. The test works both on non muscle as well as muscle invasive bladder cancer. The subtypes correlates to tumor stage and can in combination with used risk scoring tools improve the risk score by assisting with the difficult tasks to consistently define tumor stage and grade. Gene expression-based subtyping into five molecular subtypes:
- Basal squamous-like (Ba/Sq)
- Genomically Unstable (GU)
- Mesenchymal-like (Mes-like)
- Small cell Neuroendocrine-like (Sc/NE)
- Urothelial-like (Uro)
More information on Bladder Cancer.